Home
/
medtech
/
regulated medical device IVD software services
SOLUTIONS
What we do
Idneo Medical Device Software and SaMD Solutions
We specialize in developing cutting-edge software that enhances the functionality of medical devices and offers stand-alone software-as-a-device (SaMD) solutions. Our goal is to empower healthcare professionals to support clinical decision making they need to deliver superior patient care through accuracy, efficiency and real-time data.
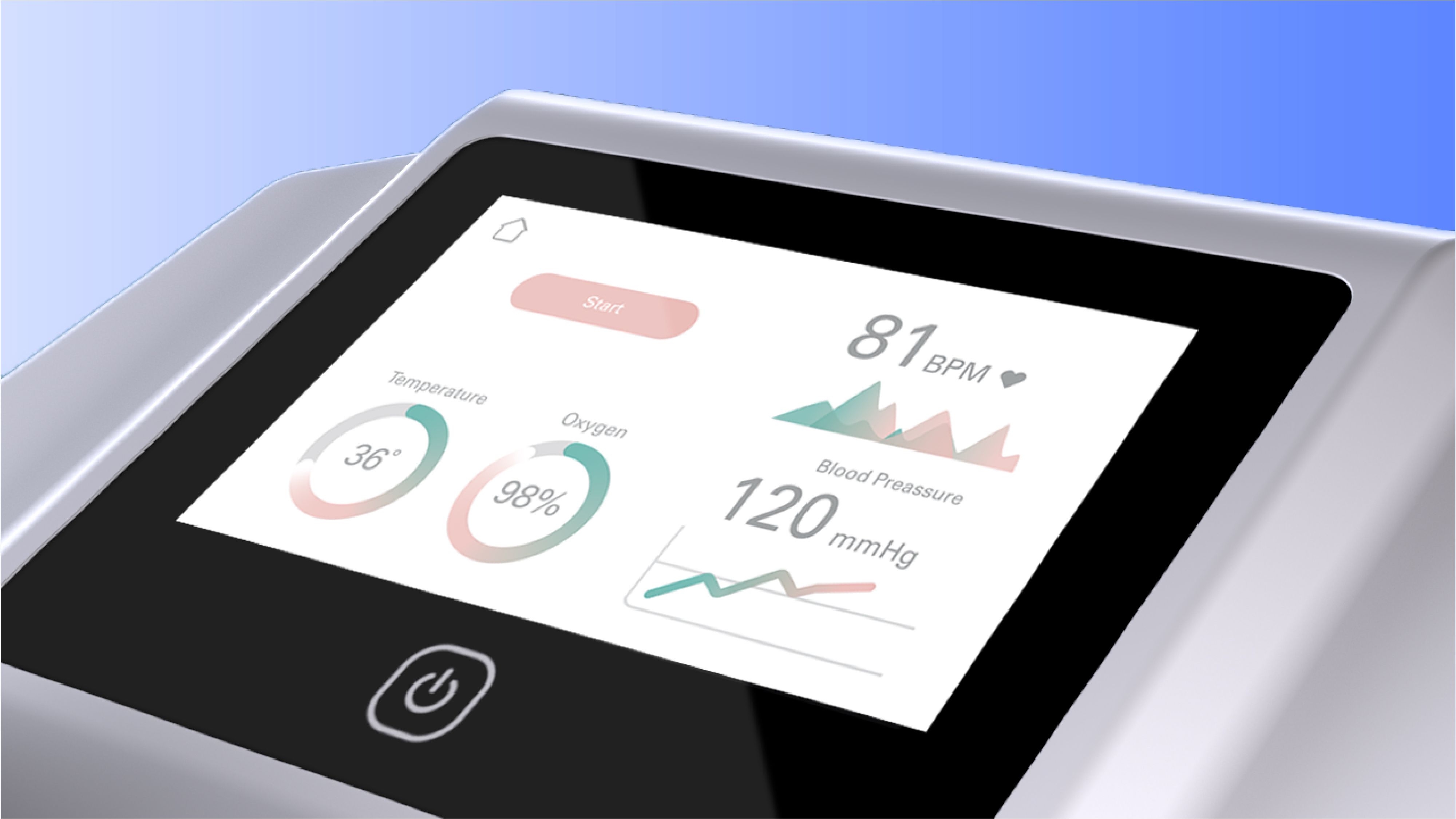

Software in a Medical Device
- Embedded in, or accessory to, a medical or In Vitro Diagnostics physical device.
- Subject to the MDR/IVDR/FDA regulations that apply to the device as a whole.
- Includes low-level firmware that controls the hardware, software that supports the device functionality, GUI through which users operate the device etc. Tools to support technical service, manufacturing QC etc may also fall in this category.

Software as a Medical Device (SaMD)
- Intended to be used for medical purposes without being part of a physical medical or in-vitro diagnostics device.
- Subject to MDR/IVDR/FDA regulations as a standalone medical device.
- Spans a broad range of applications including Clinical Decision Support Systems, Imaging Diagnostics Software, Mobile Health Apps, Patient Remote Monitoring, Telehealth Apps, etc.
CAPABILITIES
How we do it
Our software solutions integrate seamlessly with various medical devices, transforming them into intelligent systems that provide enhanced functionality and improved patient outcomes. Our broad range of software services include:

CAPABILITIES
Control application for Biochemistry analyzer
...
Pre-analytical anomaly detection using AI
...
2-channel audiometer software
...