2021
Industrial Design · User Interface Design
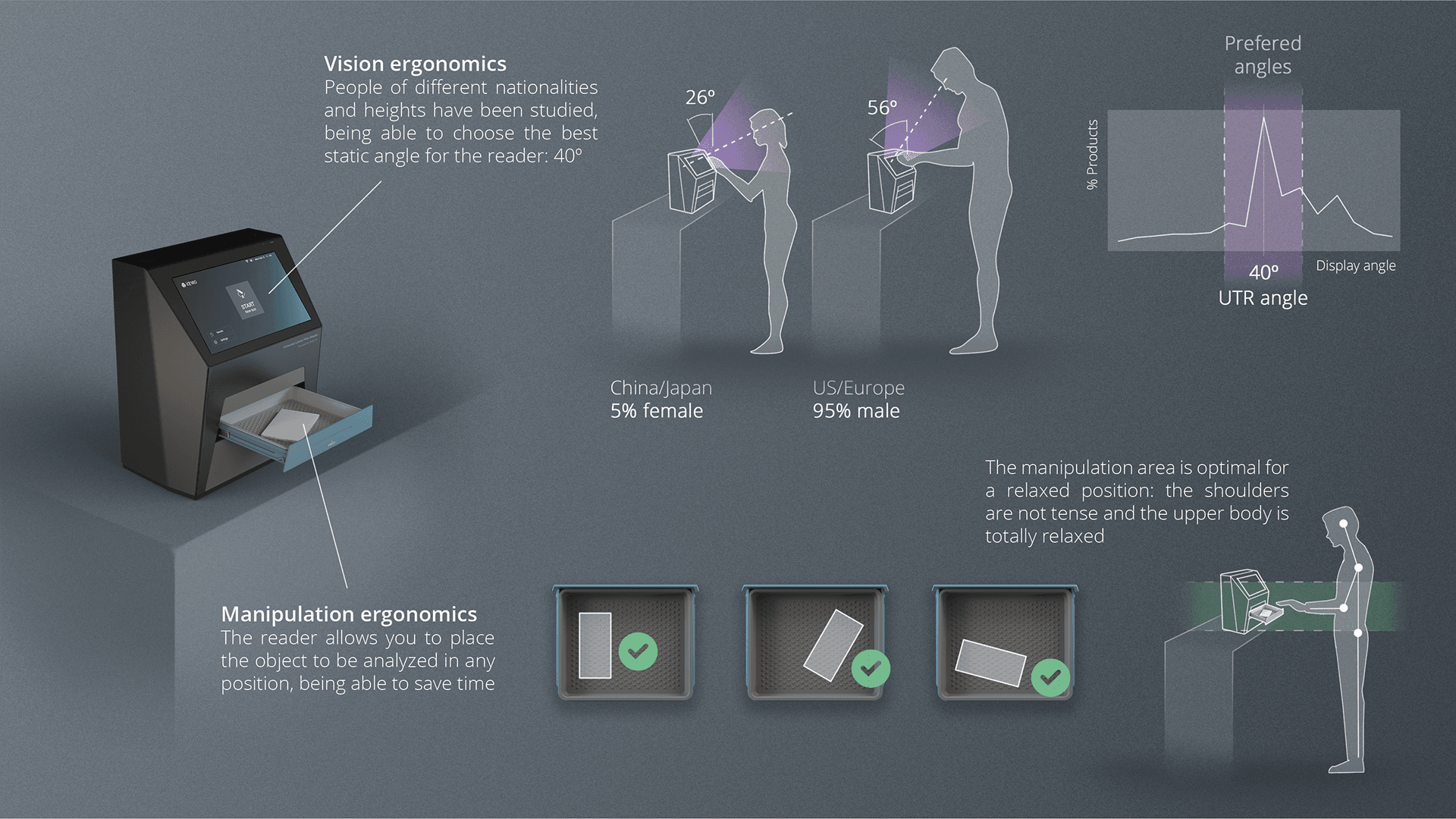
The Universal Test Reader (UTR) is Idneo Medtech’s Point of Care solution that extends the functionality of conventional rapid test reader devices by developing the innovative vision of an AI-powered open test device able to interpret rapid tests from multiple vendors and formats. The UTR addresses analytical, epidemiological and market needs, combining the quick, simple, inexpensive characteristics of rapid tests with the analytical robustness of more automated diagnostic platforms. Furthermore, it provides integrated support for managing Covid-19 use cases for pandemics control, epidemiological studies, vaccination passport management and others.

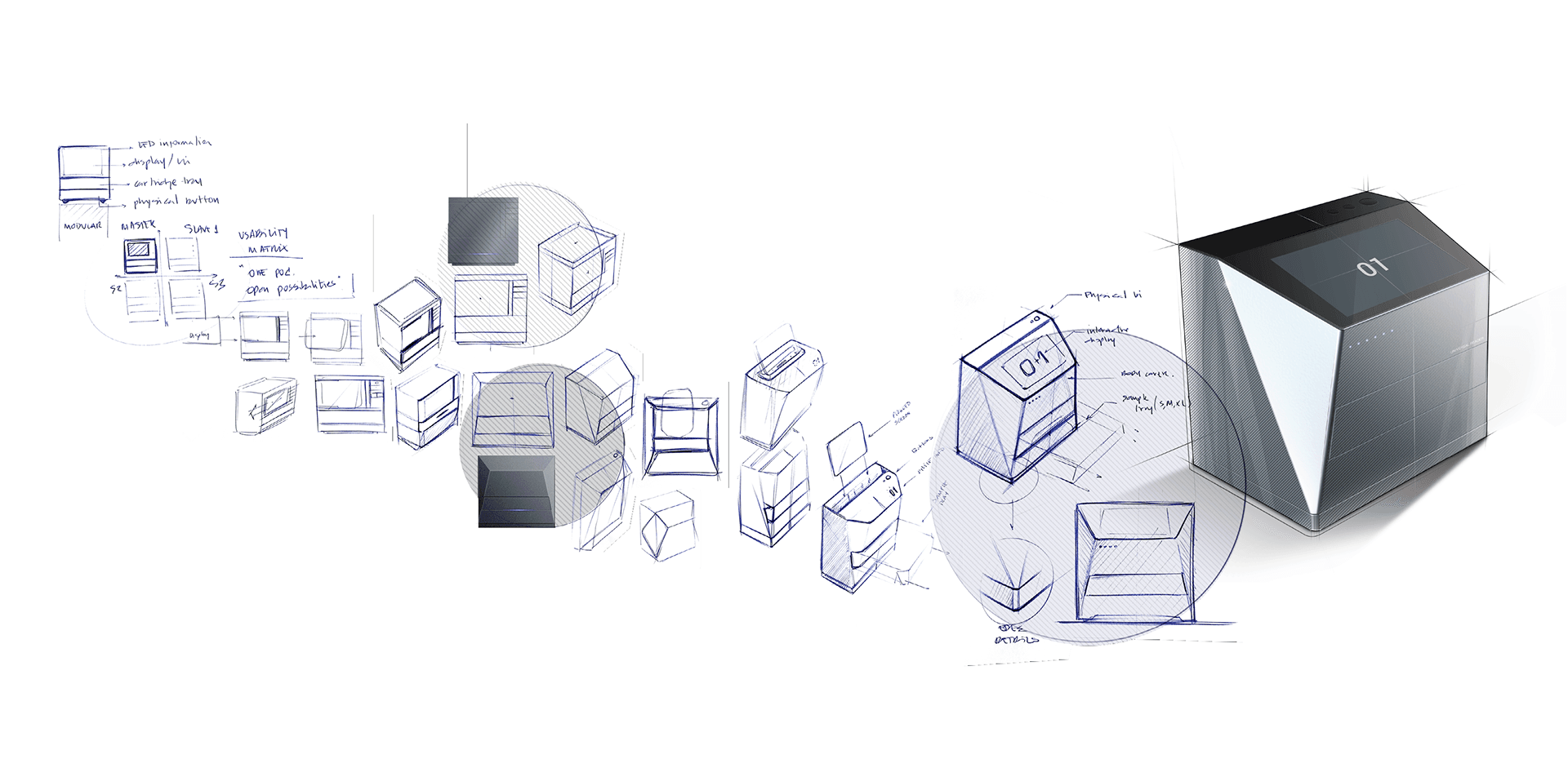
This technology is packaged in a sober design
that seeks a timeless appearance with the simplicity of
its lines and at the same time acquires its own identity